Research project
PROPER: ‘Near-patient’ prostate cancer models for the assessment of disease prognosis and therapy
How to identify patients at risk of developing devastating, metastatic disease and facilitate the development of personalised treatment for prostate cancer patients?
- Duration
- 2015 - 2019
- Contact
- Ewa Snaar-Jagalska
- Funding
-
 Alpe d’HuZes/KWF program of the Dutch Cancer Society
Alpe d’HuZes/KWF program of the Dutch Cancer Society
- Partners
Prof. dr. Gert Storm, Utrecht University
Prof. dr. Jack Schalken, Radboud University Medical Centre
Dr. Wytske van Weerden, Erasmus Medical Centre
Dr. A.T. Collins, University of York
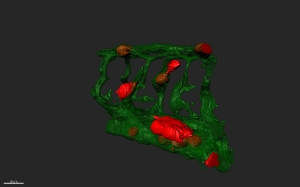
In contrast to other cancer fields, personalised medicine for patients suffering from advanced prostate tumours is still in the developmental phase. In a multi-centre approach, our expert consortium will use state-of-the-art ‘near-patient’ prostate cancer models to predict tumour aggressiveness, metastatic relapse and therapeutic response to existing and novel therapeutic compounds for individual, newly diagnosed prostate cancer patients with seemingly organ-confined disease.
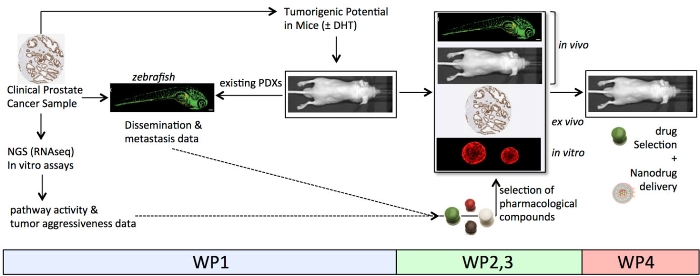
Prostate carcinoma is the most common cancer in males and the second leading cause of cancer death in the Western world. Current treatments of primary prostate tumours are initially very effective. Unfortunately, in 20-30% of patients beneficial responses are followed by tumour recurrence at distant sites leading to incurable, devastating metastatic disease. Stratification of patients at risk of developing therapy-resistant metastatic disease is, therefore, urgently needed. Moreover, current treatments of advanced prostate cancer only marginally increase overall survival and can be regarded as predominantly palliative. Hence, there is an urgent need for novel personalized therapeutic strategies for the treatment of primary tumours and for the prevention of tumour progression and metastasis formation.
The aim of our multidisciplinary consortium is to 1) better identify newly diagnosed prostate cancer patients with ostensible localized disease that are at risk of disease progression, 2) select novel, effective treatment and predict an individualized therapy response in patients (personalized medicine).
PROPER consortium consists of 6 partner institutions. It comprises 3 Dutch- & 1 UK- based prostate cancer expert groups (EMC, RUMC, LUMC, UoY) with complementary expertise in the field of pathogenesis of prostate cancer, therapy resistance, the development of new treatment for advanced prostate cancer (including nanodrug delivery), optical imaging and innovative ‘near-patient’ models of human prostate cancer. These institutions are complemented by expert
the field of nanodrug delivery (UU, Pharmaceutics) and the application of zebrafish models for applied cancer research and drug response (IBL).
The group of Ewa Snaar-Jagalska from IBL will utilize engraftment of human prostate cancer cells in zebrafish embryos to predict aggressiveness and identify candidate drug targets. In addition, IBL state-of-the-art facilities will be exploited to use the zebrafish short-term- patient-derived xenografts for rapid assessment of personalized drug response. This opens the possibility for screening on a patient-by-patient basis for drug sensitivity of tumor cells under conditions that may mimic in vivo environment.
Main contacts
Dr. Ewa Snaar-Jagalska (IBL)
Dr. Gabriël van der Pluijm (LUMC) (project coordinator)
Dr. Geertje van der Horst (LUMC)
-
Mercatali L, La Manna F, Groenewoud A, Casadei R, Recine F, Miserocchi G, Pieri F, Liverani C, Bongiovanni A, Spadazzi C, de Vita A, van der Pluijm G, Giorgini A, Biagini R, Amadori D, Ibrahim T, Snaar-Jagalska BE (2016) Development of a Patient-Derived Xenograft (PDX) of Breast Cancer Bone Metastasis in a Zebrafish Model. Int. J. Mol. Sci. 17(8): 1-11
-
Tulotta C, He S, Chen L, Groenewoud A, van der Ent W, Meijer AH, Spaink HP, Snaar-Jagalska BE (2016) Imaging of Human Cancer Cell Proliferation, Invasion, and Micrometastasis in a Zebrafish Xenogeneic Engraftment Model. Methods Mol Biol. 2016;1451:155-69. doi: 10.1007/978-1-4939-3771-4_11
-
Chen L, Groenewoud A, van der Pluijm G and Snaar-Jagalska BE (2016) A Zebrafish Xenograft Model for Studying Human Cancer Stem Cells. In press.
-
Zoni E, van der Horst G, van de Merbel AF, Chen L, Rane JK, Pelger RC, Collins AT, Visakorpi T, Snaar-Jagalska BE, Maitland NJ, van der Pluijm G (2015) miR-25 Modulates Invasiveness and Dissemination of Human Prostate Cancer Cells via Regulation of αv- and α6-Integrin Expression. Cancer Res. Jun 1;75(11): 2326-36
-
Ghotra VPS, He S, van der Horst G, Nijhoff S, de Bont H, Lekkerkerker A, Janssen R, van der Pluijm G, Jenster G, van Leenders GJLH, van de Water B, Snaar-Jagalska BE* and Danen EHJ*(2015) In vivo RNAi screen identifies SYK as a candidate drug target for prostate cancer. Cancer Res.75(1): 230-240 * corresponding authors
-
Van der Ent W, Jochemsen AG, Teunisse AF, Krens FG, Szuhai K, Spaink HP, Hogendoorn PCW, Snaar-Jagalska BE (2014) Ewing sarcoma inhibition by disruption of EWSR1-FLI1 transcriptional activity and reactivation of p53. J. Pathology, Aug;233(4): 415-24
