
The quest for more antibiotics
Streptomycetes are similar to moulds, but these bacteria live in the soil. They are very popular in biotechnology because they produce a great many antibiotics and enzymes. Gilles van Wezel will be using his Vici subsidy to study ways of increasing their production.

Foundation for Applied Sciences
‘What's special about this Vici project,' says Van Wezel, 'is that it is financed by the Foundation for Applied Sciences (Dutch: STW). This is the first time that Leiden - as a 'more general' university - has managed to attract such a subsidy, which normally goes to the technical universities. It also demonstrates the focus on biotechnology.' Van Wezel's Vici project is an example of how fundamental and applied work can go hand in hand.
Cell division
Streptomycetes produce a broad range of useful enzymes, in addition to antibiotics, which are used in the washing powder and food industries. As exotic organisms, these soil bacteria are ideally suited to study cell division. The multiplication and division of cells is a complex process. 'Streptomycetes are the only organisms where cell division is not essential, which means that mutants can easily be made and studied,' says Van Wezel. As well as fundamental reasons, he also has practical reasons for studying cell division in streptomycetes, namely to improve their growth in fermentors (incubators for micro-organisms).
Molecular switch
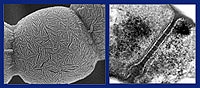
Streptomycetes have a complex lifecycle that is very similar to that of filamentous fungi, such as the fungus that makes penicillin. After the spore has germinated, a network of filaments is produced in the ground, known as the mycelium. Once the nutrients in the ground are exhausted, a white and downy airborne mycelium is formed. The airborne mycelium forms spores that are resistant to heat and drying out, which are then in turn propagated. During the development phase, the streptomycete cannibalises its own mycelium in order to acquire the growth materials it needs. As there are no more nutrients in the soil at that point in time, other antibiotics are attracted and it is probably then that antibiotics are produced to repel unwanted guests.
Signal molecule
Last year, Van Wezel c.s. clarified the mechanism that initiates antibiotic production in streptomycetes. A signal molecule plays a crucial role in this process. This molecule de-activates the regulatory protein DasR that suppresses antibiotic production. When DasR is non-active, the path is clear for production. Van Wezel: ‘We can initiate antibiotic production using a molecular switch by adding the signal molecule to a cultivation medium. What is particularly interesting is that many antibiotics that we normally do not see suddenly become visible.'
Gold mine
It is clear that this discovery constitutes a gold mine at a time when the need for new antibiotics is just as current as sixty years ago. The falling trend in discovering new antibiotics is a major problem for world health. Van Wezel also co-ordinates another project financed by STW, together with other Leiden scientists such as Erik Vijgenboom, Hermen Overkleeft, Mark Overhand, Rob Verporte and Young Choi, in which they are actively seeking hidden antibiotic clusters - the antibiotics of the future.
Knock-out mutants
Cell division in relation to the growth of streptomycetes is a key focal point in his own Vici project. The SsgA protein is crucial in the formation of cell-wall material. All streptomycetes have several SsgA-like proteins (SALPs). Van Wezel: ‘We have recently made stems which lack the information for one of the proteins, the so-called knock-out mutants. It has been shown that SALPs without exception play a role in cell division, from stimulating cell division to releasing the mature spores.'
Patented technology

The basis for this research was already laid at the end of 1996 with the request from DSM-Delft to try to have streptomycetes grow in a more fragmented fashion. In the fermentor, streptomycetes generally form endlessly long hyphae (the threads that form the mycelium) and tight networks of interwoven hyphae. Van Wezel: ‘This method of growth is very unfavourable for large-scale production. The large mycelium clumps lead to slow growth, resulting in too low a yield. SsgA strongly influences growth: the more SsgA, the more cell division and fragmentation. This makes it possible to influence the growth genetically, and by using a number of genetic tools to manipulate the process.' This patented technology has been shown to lead to a considerable improvement in yield.
‘Black box’ approach
‘We have now taken a number of steps in the right direction, but we have come up against a new problem: the relation between growth and antibiotic production. Many antibiotics need a minimum size of mycelium clump before anything can be produced, while other antibiotics and many enzymes benefit from very small fragments. This phenomenon has never been investigated. The industry generally prefers optimising production via a 'black box' approach. Nobody knows where products are made and secreted in the streptomycetes. Is it at the tips, where growth takes place, or in the middle, along the walls of the hyphae? It is important to gain an insight into this as this is the only way to achieve specific improvement of this plant group.'
Continuity
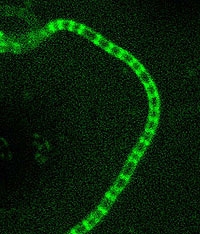
In Van Wezel's Vici project, supported by DSM and Danisco-Genencor, there are three core research themes: analysing where production and excretion take place and how they respond to growth conditions; determining how the activity of the key genes depends on growth and the rate of growth; and constructing a predictive computer model to integrate all the processes, in co-operation with Mark van Loosdrecht and Christian Picioreanu of the TU Delft, so that a digital test run of a fermentation process can be carried out and much more focused processes can be directed and improved. He will also be developing innovative technology for genetic manipulation of streptomycetes. Van Wezel: 'It's this very duality (fundamental and applied) that makes intensive co-operation between the TU Delft and Leiden University so important for the continuity of this kind of research.'
(12 May 2009)
