Tjerk Oosterkamp Lab - Microscopy and Quantum Mechanics at milliKelvin temperatures
Towards nano-MRI
By detecting the tiny forces between a micrometer sized magnet and the spins of hydrogen nuclei, we can do MRI with a volume resolution that is approximately 12 orders of magnitude better than a conventional MRI.
In our group, we work on the development of MRFM (Magnetic Resonance Force Microscopy) to image both electron spins, and nuclear spins. The imaging of these nuclear spins is not unlike what is done in conventional MRI in scheme. Our technique allows us to image far lower number of spins however, resulting in a higher resolution. This is called NanoMRI.
Compared to other scanning probe techniques, one of the main advantages of MRFM lies in where we can image: whereas techniques such as Scanning Tunnelling Microscopy (STM) or conventional Atomic Force Microscopy (AFM) can only image the top most layers of a sample, MRFM is able to image up to a micron below the surface. This allows us to image a much wider breadth of samples. More on this can be found under “MRFM in Quantum Matter”. [ TODO this section is not yet written, link to it when it is]
Another advantage lies in the non-invasive nature of the technique. Whilst techniques such as Electron Microscopy and X-ray Diffraction can also be used to penetrate the surface layer of samples, the highly energetic particles used in such techniques are potentially damaging to the sample being studied. This is not the case for NanoMRI using MRFM, where the sample will not “feel” much of the imaging. This means that the exact same sample can be imaged repeatedly and for longer periods of time.
The combination of these characteristics makes NanoMRI very suited for 3D biological imaging. The current state of the art within MRFM lies in the 3D imaging of the Tobacco mosaic virus with a resolution of several cubic nanometers. Improving the technique towards atomic resolution imaging would open the possibility of imaging, for example, proteins and DNA in 3D, giving us a whole new insight into their working, and a new way of experimentally verifying many models on the 3D structure of these more complex compounds and their chemistry at the basis of our biology.
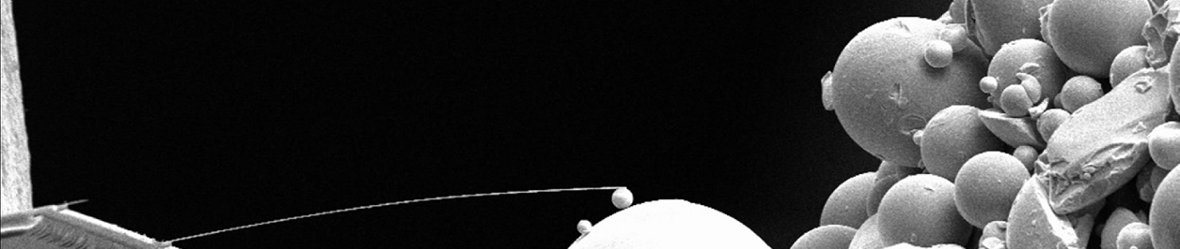
To improve this resolution, we are currently working on augmenting our force sensing cantilevers with a double magnetic tip. Using a larger magnetic particle for our SQUID readout, and a smaller particle with a resultant high magnetic field gradient for sensing allows us to take thinner resonant slices, and thus increase the resolution of our technique.
Another project we are currently working on, is to directly image perodically patterned samples containing protons. For this we use a substrate with a certain spatial periodicity, coated with a highly protonated sample. This will help us quantify our resolution using our current state of the art and evaluate future improvements.