Research project
Nanoparticle – redox protein biohybrids
Artificial photosynthesis aims to produce fuels from solar energy using chemical processes. In semi-artificial photosynthesis, a hybrid approach is taken using both chemical and biotechnology approaches. We aim to develop hybrid systems between light-harvesting nanoparticles and redox-enzymes (oxidoreductases) and study the interaction and electron transfer properties between the components. We study our biohybrids systems using a range of photochemical, biophysical and bioelectrochemical methods. Our ultimate vision to create biohybrid systems between nanoparticle photocatalysts and whole-cell biocatalyst (bacteria) in which solar light is harvested by the nanoparticles and one of the two electrocatalytic half-reactions is performed in by the whole-cell biocatalyst.
- Contact
- Lars Jeuken
In photosystem II, light energy is transfer to the special pair (P680), where it excites an electron that is subsequently transferred across a chain of redox-active co-factors. The hole that is left at the P680+ is filled by electrons released at the oxygen-evolving centre (OEC) where water is oxidised. Although this system is one of the most quantum efficient systems known, photosystem II is prone to photo-oxidative damage, which can limit plant growth and impairs exploitation in biotechnology. In semi-artificial photosynthesis, our aim is to biomimic natural photosynthesis with more robust light-harvesting nanoparticles. To retain the high-activity and specificity of chemical transformation in nature, the nanoparticles are coupled to biocatalyst (e.g. enzymes) for energy conversion. This means that electrons have to transfer from the light-harvesting nanoparticles to the biocatalyst and because electron transfer rates exponentially decrease with distance, the nanoparticle needs to be engineered to be very close to the redox-active co-factors of proteins (<2 nm).
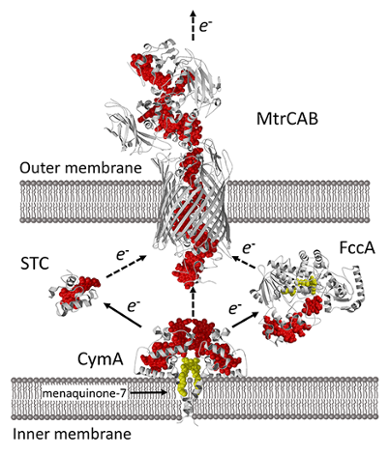
Nanoparticle – MtrCAB biohybrids for whole cell biocatalysis
Biohybrid systems that use whole-cell biocatalyst like bacteria would be able to exploit the regenerative capabilities of the living cell. For this, electrons need to transfer from the nanoparticle into the periplasm or cytoplasm of the whole-cell biocatalyst. In collaboration with Prof. Butt from the University of East Anglia and Prof. Reisner from the University of Cambridge, we are studying whether this can be achieved using the metal-reducing pathway in Shewanella oneindensis. Key to this pathway is the protein complex, MtrCAB, which sits in the outer membrane and transports respiratory electrons from the periplasm to the extracellular environment. In this project we are studying whether we hijack this pathway to exchange photoelectrons with S. oneidensis.
Key publications
Zhang H., Jaenecke J., Bishara-Robertson I.L., Casadevall C., Redman H.J., Winkler M., Berggren G., Plumere N., Butt J.N., Reisner E. & Jeuken L.J.C. (2024), Semiartificial photosynthetic nanoreactors for H2 generation, Journal of the American Chemical Society 146(50): 34260-34264. DOI: 10.1021/jacs.4c12311
Robertson I.L., Zhang H., Reisner E., Butt J.N. & Jeuken L.J.C. (2024), Engineering of bespoke photosensitiser-microbe interfaces for enhanced semi-artificial photosynthesis, Chemical Science 15(26): 9893-9914. DOI: 10.1039/D4SC00864B
van Wonderen, J.H., Adamczyk, K., Wu, X., Jiang, X., Piper, S.E.H., Hall, C.R., Edwards, M.J., Clarke, T.A., Zhang, H., Jeuken, L.J.C., Sazanovich, I.V., Towrie, M., Blumberger, J., Meech, S.R. and Butt, J.N. (2021) Nanosecond heme-to-heme electron transfer rates in a multiheme cytochrome nanowire reported by a spectrally unique His/Met-ligated heme, Proc. Nat. Acad. Sci. USA, 118, e2107939118. DOI: 10.1073/pnas.2107939118
van Wonderen, J.H., Adamczyk, K., Wu, X., Jiang, X., Piper, S.E.H., Hall, C.R., Edwards, M.J., Clarke, T.A., Zhang, H., Jeuken, L.J.C., Sazanovich, I.V., Towrie, M., Blumberger, J., Meech, S.R. and Butt, J.N. (2021) Nanosecond heme-to-heme electron transfer rates in a multiheme cytochrome nanowire reported by a spectrally unique His/Met-ligated heme, Proc. Nat. Acad. Sci. USA, 118, e2107939118. DOI:10.1073/pnas.2107939118
Stikane, A., Hwang, E.T., Ainsworth, E.V., Piper, S., Critchley, K., Butt, J.N., Reisner, E., Jeuken, L.J.C., (2019) Towards compartmentalized photocatalysis: Multiheme proteins as transmembrane molecular electron conduits, Faraday Discussion, 215, 26-38. DOI:10.1039/C8FD00163D
Hwang, E.T., Sheikh, K. Orchard, K.L., Hojo, D, Radu, V., Lee, C.-Y., Ainsworth, E., Lockwood, C., Gross, M.A., Adschiri, T., Reisner, E., Butt, J.N. and Jeuken, L.J.C. (2015) A Decaheme Cytochrome as a Molecular Electron Conduit in Dye-Sensitized Photoanodes, Adv. Funct. Mater. 25, 2308-2315. DOI: 10.1002/adfm.201404541
