Research project
Inverse Agonism and Constitutive Activity
-
- Contact
- Laura Heitman
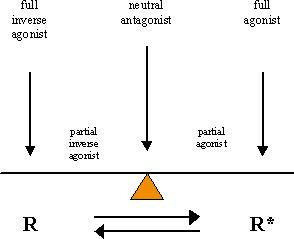
It has been found that receptors may exist in a constitutively active state, particularly when expressed in high amounts in cultured cells. Such receptors, "active on their own", may be calmed down by so-called inverse agonists, often compounds that were originally classified as antagonists. To explain all this one has to accept that a receptor can exist in (at least) two states, one inactive (R), the other active (R*). Agonists, such as hormones, neurotransmitters and some drugs (e.g., salbutamol and terbutaline against asthma), prefer and select the active state R*, while inverse agonists go for R. In this concept "neutral" antagonists would not alter the R/R* distribution (see Figure).
Capital question, of course, is whether this phenomenon is of relevance in pathophysiological conditions or rather a pharmacological curiosity. We have worked together with leading European academic groups to address this question and to study the concept of inverse agonism through a variety of methodologies ranging from organic chemistry to molecular biology.
We learned the following:
- inverse agonism is drug and system dependent; though the system may not allow for the detection of negative intrinsic efficacy
- in reality, many antagonist drugs on the market are actually inverse agonists
- structure-activity relationships for inverse agonists are emerging
- constitutively active receptors are inherently instable
- it may well be that most wild-type receptors are relatively silent; upon mutation they may become (constitutively) active – these mutations seem quite random
If you'd like to know more about this subject, we wrote two comprehensive and well-cited reviews quite some years ago. Here are the references:
- R.A.F. de Ligt, A.P. Kourounakis, A.P. IJzerman (2000) Inverse agonism at G protein-coupled receptors: (Patho)physiological relevance and implications for drug discovery. Br. J. Pharmacol. 130: 1-12
- R.A. Bond, A.P. IJzerman (2006) Recent developments in constitutive receptor activity and inverse agonism, and their potential for GPCR drug discovery. Trends Pharmacol. Sci. 27: 92-96.
We also wrote a review article on the structure-activity relationships of inverse agonists:
- W. Soudijn, I. Wijngaarden, A.P. IJzerman (2005) Structure-activity relationships of inverse agonists for G-protein-coupled receptors. Med Res Rev. 25: 398-426.
Well, we’re not only writing reviews on this topic. We now use a yeast system in which we can express and mutate receptors very easily. Upon activation of the receptors the yeast cells start growing, which, of course, is a very easy read-out. So simple in fact that we can screen thousands of mutants. This is extremely informative, as we learned that many mutants are constitutively active, and these mutations pop up at very different locations in the receptor. This is very puzzling: it seems to contradict a very precise mechanism of receptor activation. If you want to know more:
- Liu R, Groenewoud NJ, Peeters MC, Lenselink EB, IJzerman AP. A yeast screening method to decipher the interaction between the adenosine A2B receptor and the C-terminus of different G protein α-subunits. Purinergic Signal. 2014, 10(3):441-453.
- Peeters MC, Wisse LE, Dinaj A, Vroling B, Vriend G, IJzerman AP. The role of the second and third extracellular loops of the adenosine A1 receptor in activation and allosteric modulation. Biochem Pharmacol. 2012, 84(1):76-87.
- Peeters MC, Li Q, van Westen GJ, IJzerman AP. Three "hotspots" important for adenosine A2B receptor activation: a mutational analysis of transmembrane domains 4 and 5 and the second extracellular loop. Purinergic Signal. 2012, 8(1):23-38.
- Peeters MC, van Westen GJ, Li Q, IJzerman AP. Importance of the extracellular loops in G protein-coupled receptors for ligand recognition and receptor activation. Trends Pharmacol Sci. 2011, 32(1):35-42.
- Q. Li, K.Ye, C.C. Blad, H. den Dulk, J. Brouwer, A.P. IJzerman, M.W. Beukers (2007) ZM241385, DPCPX, MRS1706 are inverse agonists with different relative intrinsic efficacies on constitutively active mutants of the human adenosine A2B receptor. J Pharmacol Exp Ther. 320: 637-645
- M.W. Beukers, J. van Oppenraaij, P.P. van der Hoorn, C.C. Blad, H. den Dulk, J. Brouwer, A.P. IJzerman (2004) Random mutagenesis of the human adenosine A2B receptor followed by growth selection in yeast. Identification of constitutively active and gain of function mutations. Mol Pharmacol. 65: 702-710.
