Research project
Intracellular allosteric modulators for human CC chemokine receptors
Supervisor: Natalia Ortiz Zacarías
- Contact
- Daan van der Es
 The chemokine system is comprised of more than 20 different chemokine receptors—which belong to the class A or rhodopsin-like family of GPCRs—and almost 50 chemokine ligands that play a critical role in the immune system. Dysregulation of this system can lead to different pathologies, including inflammatory and autoimmune diseases such as atherosclerosis, rheumatoid arthritis and even cancer. The involvement of these receptors in many pathologies has resulted in many efforts to develop chemokine receptor antagonists; however, only a few antagonists have been approved for clinical use so far. In general, lack of efficacy has been reported as the main reason of Phase II and Phase III failures, making the development of more predictive preclinical models in earlier phases of research highly necessary. A thorough understanding of the mechanism of action at a molecular level is crucial for the development of drug candidates with better safety and efficacy profiles. In this regard, the concepts of drug-target residence time and allosteric modulation have gained increased interest as more evidence suggests that they play a critical role in in-vivo efficacy and safety.
The chemokine system is comprised of more than 20 different chemokine receptors—which belong to the class A or rhodopsin-like family of GPCRs—and almost 50 chemokine ligands that play a critical role in the immune system. Dysregulation of this system can lead to different pathologies, including inflammatory and autoimmune diseases such as atherosclerosis, rheumatoid arthritis and even cancer. The involvement of these receptors in many pathologies has resulted in many efforts to develop chemokine receptor antagonists; however, only a few antagonists have been approved for clinical use so far. In general, lack of efficacy has been reported as the main reason of Phase II and Phase III failures, making the development of more predictive preclinical models in earlier phases of research highly necessary. A thorough understanding of the mechanism of action at a molecular level is crucial for the development of drug candidates with better safety and efficacy profiles. In this regard, the concepts of drug-target residence time and allosteric modulation have gained increased interest as more evidence suggests that they play a critical role in in-vivo efficacy and safety.
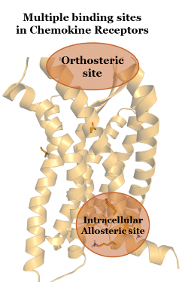
Recently, it was discovered that the CC chemokine receptor 2 (CCR2) and other chemokine receptors (such as CCR4, CCR5 and CXCR2) possess multiple binding sites, allowing for its modulation by so-called orthosteric and allosteric antagonists. Allosteric antagonists represent an important therapeutic option as, unlike orthosteric antagonist, they are able to act in a non-competitive manner. This makes them highly effective even in high local concentration of chemokines. Moreover, this intracellular binding site is highly-conserved among several chemokine receptors, allowing the development of a multiple-target approach, for example dual intracellular antagonists. In addition, inclusion of residence time in early hit-to-lead optimization has been recently used for the successful development of high-affinity and long-residence time orthosteric antagonists of the human Chemokine Receptor 2 (CCR2). However, inclusion of residence time in the development of novel allosteric modulators is still missing. With these objectives, we aim to develop new insight on the intracellular allosteric binding site of chemokine receptors, specifically CCR1 and CCR2, as this knowledge might lead to the development of insurmountable antagonists with improved in-vivo outcomes.
Techniques:
- Cell culture
- Radioligand binding assays
- Functional assays
