Research project
From stress to success: how actinobacteria exploit life without a cell wall
The central question of this Vici proposal is to investigate if, and how actinobacteria exploit life without a cell wall.
- Duration
- 2020 - 2025
- Contact
- Dennis Claessen
- Funding
-
 NWO Vici
NWO Vici
Description
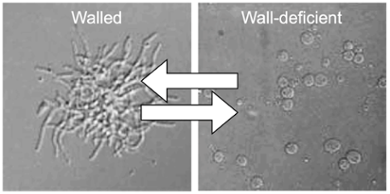
The cell wall is a conserved structure in bacteria, which protects cells from environmental stresses. Given its protective role, the cell wall is the target for many antibiotics. My team recently discovered that some actinobacteria, paradoxically, have a natural ability to shed their cell wall under influence of stress, yielding cells that are wall-deficient. This wall-deficient state is not permanent, allowing cells to revert to their canonical walled mode-of-growth. Although wall-deficient cells are intrinsically fragile, this fragility is offset with crucial benefits, which have so far not been investigated. For instance, wall-deficient cells are malleable, which facilitates dispersal of cells inside hosts via narrow spaces that are inaccessible to walled cells. Furthermore, given that wall-associated epitopes are often recognized by host defence systems, wall-deficiency provides a plausible explanation for how pathogens may hide in their host and cause recurrent infections. Finally, wall-deficient cells are resistant to wall-targeting antibiotics and proficient at DNA uptake and exchange, which may foster horizontal gene transfer and the spread of antimicrobial resistance. Taken together, these examples demonstrate that wall-deficiency provides cells with various capabilities that are unavailable to walled cells.
The aim of this Vici proposal is to dissect how actinobacteria benefit from a wall-deficient lifestyle. Through state-of-the-art technologies including transposon sequencing, microfluidics approaches and high-end imaging I will investigate which actinobacteria can switch between the walled and wall-deficient state and how these transitions are mechanistically regulated. I will study how wall-deficiency allows actinobacteria to manoeuvre through host tissue and to rapidly evolve new beneficial traits. These integrative approaches will substantially advance our understanding of the functional and evolutionary consequences of morphological plasticity in bacteria. The results obtained in this project can be used to improve diagnostics in clinical settings and develop effective treatment strategies to combat devastating pathogens that exploit this extreme form of adaptation.