Nose of E. coli zips open and shut
PhD student Wen Yang discovered how certain cell receptors in E. coli bacteria signal 'smells'. With the use of ice-cold electron microscopy microbiologists from Leiden gain more insight into how bacteria respond to their environment. Publication in mBio.
Fight infection diseases
Bacteria use chemotaxis to respond to their environment. They move as a result of the concentration of certain substances in their surroundings, for example to search for food sources or to leave an unfavorable environment. Prof.dr. Ariane Briegel, involved in this research, wants to know more about the 'nose' of bacteria: 'Chemotaxis plays an important role in the first steps of host invasion for pathogenic bacteria. Understanding it might help in the development of new antimicrobial agents.' With this new insight Briegel and her team have now learned more about how the signal travels from the 'nose' to the 'tail' (flagellum) that allows bacteria to move in a targeted manner.
Shock freeze
For their discovery PhD student Wen Yang and her colleagues used a cryo-electron microscope at the Netherlands Centre for Electron Nanoscopy (NeCEN) in Leiden. Inside the microscope the temperature is almost 200°C below zero. By freezing microbes very quickly they are pristinely preserved in a glass like ice. With the use of cryotomography the researchers are able to resolve the microbes in 3D and in more detail than a usual electron microscope would allow. The team led by professor Briegel has taken hundreds of photos of a harmless strain of the Escherichia coli bacterium containing different activation stages of the receptors that ‘smell’ their chemical environment.
Zip it up
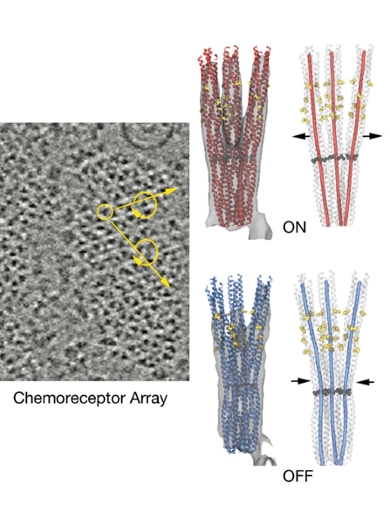
The chemoreceptors that form the 'nose' of the E. coli are only a few nanometers in size and are arranged in a highly ordered hexagonal lattice pattern, like a honeycomb (image left). Receptors (images right) protrude from the bacterium envelope into the inside of the cell. They form a connection between the inner world of the bacterium and the environment. The researchers have discovered that depending on the type of molecules that bind on the outside, a certain amino acid hinge allows the receptor to zip up or be in a more open state (images top and bottom right), while maintaining the hexagonal structure. This allows for signal propagation in the bacterium with the aim of controlling the movement of the E. coli.
Image at the top: E. coli has several 'tails' to propel itself with (artist impression). Illustrators: Alissa Eckert and Jennifer Oosthuizen / Content by CDC
