
White blood cells in transparent embryos
Leiden molecular cell biologists in the research group of Annemarie Meijer have discovered novel early macrophage-specific genes in zebrafish, including a signal transducer pivotal for the migration of macrophages in the innate immune response to bacterial infection. Their findings were published on April 27 in the medical journal Blood, with postdoc Anna Zakrzewska and PhD student Chao Cui as shared first authors of the paper.
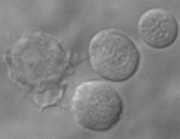
Macrophages are white blood cells that are crucial for the immune response to microbial infections. Macrophages rapidly migrate to infection sites and efficiently engulf and destroy bacteria or other invading microorganisms. They also function to clear the body of cellular debris, and damaged or senescent cells. In all vertebrate animals, the development of macrophages and other white blood cells is controlled by a transcription factor that is known as Spi1 or Pu.1. However, despite extensive studies of Spi1, it remains largely unknown which precise gene groups are regulated by this essential transcription factor.
The zebrafish model
To identify immune response genes that are dependent on the essential Spi1 transcription factor, the Leiden group made use of the zebrafish embryo model. Zebrafish embryos already possess an innate immune system at the first day of their development. Since the embryos are optically transparent, they are ideal to study the development and function of immune cells. In addition, very efficient gene knock-down technology is available to study gene functions in the zebrafish embryo model.
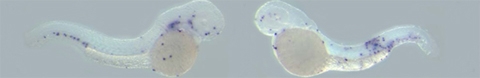
Gene discovery
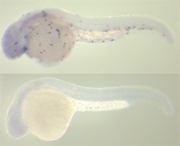
For the discovery of novel macrophage markers in zebrafish a microarray strategy was used. This technique allows simultaneously interrogating the expression of many thousands of genes. Gene expression in control embryos was compared with gene expression in embryos that lacked macrophages due to knock-down of the Spi1 transcription factor that is required for their development. In addition, a transgenic zebrafish line was used in which the early precursors of white blood cells are marked by expression of Green Fluorescent Protein (GFP). The GFP-positive cells were isolated from embryos of this transgenic line by a technique called Fluorescence Activated Cell Sorting. By determining the overlap between genes down-regulated upon Spi1 knock-down and genes enriched in the GFP-positive cell fraction, a large set of Spi1-dependent genes that are expressed in cells of the embryo’s immune system could be identified.
Expression patterns
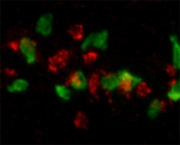
The identified gene group contained several known genes with roles in blood cell and immune system development as well as novel genes with immune-related functions. The next step was to determine the specificity of the expression of these genes in different cell types of the immune system, such as macrophages and neutrophils. Fluorescent co-localization experiments with known markers for these cell types revealed several novel genes that are expressed in macrophages during early stages of embryo development. These genes are now being used to develop new transgenic lines that will enable real-time imaging of macrophage behavior in living embryos.
Cell migration to infection sites
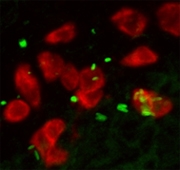
One of the early macrophage-specific genes codes for a chemokine receptor called cxcr3.2. Chemokine receptors are signal transducers that sense chemokines, which are small secreted proteins that act as chemoattractants to guide the migration of immune cells. To study the function of this receptor a knock-down analysis was performed. Salmonella bacteria were locally injected into the tail muscle of embryos to study immune cell migration under cxcr3.2 knock-down conditions. These studies showed that cxcr3.2 is pivotal for the migration of macrophages during the innate immune response of zebrafish embryos to bacterial infection.
Links
- The article Macrophage-specific gene functions in Spi1-directed innate immunity. Blood. 2010 Apr 27. [Epub ahead of print].
