Research project
COMMUNITY: unraveling the regulatory networks in Streptomyces that switch on antibiotic production on demand
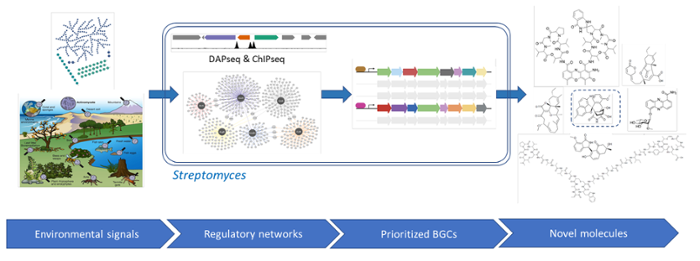
Through his project we will unravel the global regulatory networks that control gene expression in Streptomyces bacteria and allow them to properly respond to major changes in the environment; we will then harness this knowledge to activate and identify novel antibiotics
- Duration
- 2022 - 2027
- Contact
- Gilles van Wezel
- Funding
-
 ERC Advanced grant
ERC Advanced grant
- Partners
Marnix Medema, Wageningen University
Jos Raaijmakers, NIOO-KNAW
Description
As Nature's medicine makers, Streptomyces bacteria produce a plethora of natural products, which we harness for clinical, biotechnological and agricultural applications, including 70% of the antibiotics. Streptomycetes still have a vast reservoir of unexplored biosynthetic potential, but many biosynthetic gene clusters (BGCs) are not expressed in the laboratory. To bring the chemical dark matter to the light, we need to discover the keys to unlock the expression of cryptic BGCs. Aim is to add a new dimension to genome mining, via understanding and exploitation of the regulatory networks that control natural product biosynthesis in Actinobacteria. My team discovered the concept of antibiotic production on demand, showing that plant hormones activate antimicrobials. Predicting when instead of what BGCs produce will allow clustering of BGCs based on their response to ecological signals. This can serve as a beacon for prioritising BGCs, and aid in the discovery of new biosynthetic pathways. I will tackle three major challenges:
- The systems biology challenge is to elucidate the regulatory circuitry of streptomycetes and to reliably predict how BGCs are controlled.
- The metabolic challenge is to unwire the networks that tie carbon metabolism to antibiotic production, to bridge the gap from the complex polysaccharides in nature to the defined carbon sources of the laboratory.
- The ecological challenge is to unravel the mechanisms and molecules via which plants invoke the power of Streptomyces' bioactive molecules to obtain protection against infections and pests, aimed at biological disease-suppression.
COMMUNITY is an open science project that will help to elucidate whether the yet unexplored BGCs will deliver a paradigm shift in drug discovery, for application in agriculture and human health. Deliverables are innovative systems biology tools and detailed transcription factor networks, elicitors for drug screening and disease-suppressive microbes