Research project
Cell architecture and pathways for parallel secretion in the filamentous fungus Aspergillus niger
Research aims: Identification of key genes involved in programming the cellular architecture of A. niger & Genetic engineering of A. niger in order to improve its secretory capacities and rheological behavior under industrial fermentation conditions.
- Duration
- 2009 - 2014
- Contact
- Arthur Ram
- Funding
-
 Kluyver Centre for Genomics of Industrial Fermentation (KC)
Kluyver Centre for Genomics of Industrial Fermentation (KC)
- Partners
Han Wosten - University Utrecht
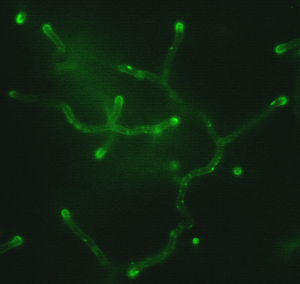
Cell morphology and secretion capacity are two important parameters in relation to optimizing enzyme production. Fundamental research on fungal morphology and secretion in the industrial host Aspergillus niger is aimed at a much deeper understanding of the mechanisms that temporally and spatially regulate different steps of the hyphal morphogenesis and secretion. Knowledge of the complex networks and molecular interactions will generate new possibilities to control and manipulate the morphology of A. niger for industrial applications.
The filamentous fungus Aspergillus niger is an industrially important fungus commonly exploited for the production of enzymes and organic acids. In this project we study how morphology is involved in several cellular processes.
Analysis of protein secretion in A. niger suggested that proteins are mainly secreted from hyphae at actively growing tips, implying that protein secretion and hyphal tip growth are coupled processes. Thus, the polar growth mode of A. niger is assumed to be one of the key parameters that determines product yields. Although there have been several attempts to relate the productivity of Aspergillus cultivations with morphological characteristics, no generally accepted model exists so far that can be used as basis for rationally optimising the morphology of A. niger with respect to its behaviour in a bioreactor and protein production/secretion.
To ascertain the set of gene products involved in the cellular architecture and secretion of A. niger, i.e. in the establishment and maintenance of polarity, different complementary approaches will be followed. Comparative genome-wide expression profiling studies using a defined morphological mutant as well as wild type strains induced for morphological alterations will be performed. We will aim to elucidate the molecular mechanism(s) which enables A. niger to increase its protein producing capacities at identical growth rates and construct strains in which growth and protein secretion are uncoupled allowing more protein to be produced with less biomass.
- Meyer V, F Wanka, J van Gent, M Arentshorst, CA van den Hondel, AF Ram. 2011. Fungal gene expression on demand: An inducible, tunable and metabolism independent expression system for Aspergillus niger. Appl. Environ. Microbiol. 77:2975-2983.
- Kwon MJ, M Arentshorst , ED Roos, CA van den Hondel, V Meyer, AF Ram. 2011. Functional characterization of Rho GTPases in Aspergillus niger uncovers conserved and diverged roles of Rho proteins within filamentous fungi, Mol. Microbiol. 79:1151-1167
- Kwon MJ, Jørgensen TR, Nitsche BM, Arentshorst M, Park J, Ram AF, Meyer V. 2012. The transcriptomic fingerprint of glucoamylase over-expression in Aspergillus niger. BMC Genomics. 13:701.
- Kwon MJ, Nitsche BM, Arentshorst M, Jørgensen TR, Ram AF, Meyer V. 2013. The transcriptomic signature of RacA activation and inactivation provides new insights into the morphogenetic network of Aspergillus niger. PlosOne; 24;8 (7): e68946
- Kwon MJ, Arentshorst M, Fiedler M, de Groen FL, Punt PJ, Meyer V, Ram AF. 2014. Molecular genetic analysis of vesicular transport in Aspergillus niger reveals partial conservation of the molecular mechanism of exocytosis in fungi. Microbiology (SGM), 160: 316-329.
