
Recycling at microscale
Playing with tiny building blocks might sound like child’s play, but Vera Meester knows better. On June 7 she will defend her thesis on colloids: micro particles with which you can form larger structures. Meester developed a method to make unusable structures usable.
Visible microparticles
Colloids can consist of all kind of substances and are present in many everyday products, like food, medicines and cosmetics. They determine the taste, texture and appearances of these products. The particles are no larger than a micrometer, but still visible with a microscope. ‘That is the real fun part of those colloids,’ says Meester. ‘It allows you to study them in real time and immediately see how the particles behave in a liquid. This is different with, for example, atoms or nanoparticles, which can often only be observed indirectly with the help of measurements.’ The particles that Meester used to work on consisted of plastic.
Lego 2.0
Meester explains that she can adapt the chemical and physical properties of the colloids. By changing the interaction between or the shape of the particles, for example, colloids can act as building blocks that self-assembly into larger structures. ‘The ultimate goal is that you get structures like Lego blocks that by themselves build a house or a car, depending on what kind of building blocks you use,’ Meester goes on. This is why she studied the behaviour and properties of the colloids and made different particles and structures herself. ‘We have carefully examined which building blocks we can make, why they are made in this way and how we can functionalise them.’
Smart materials
This fundamental understanding of the behaviour and properties of colloids is necessary to develop new materials with specific properties. Complex building blocks mean you can make smart materials. These materials adapt their structure to the environment. By changing the temperature or pH, you can make such structures expand or shrink, for example.
Making building blocks
Meester made the building blocks by turning spherical particles into anisotropic particles. ‘Anisotropic means: everything that is not spherical,’ she explains. ‘By adding chemicals, I changed the roughness of the surface and created indentations in the surface of the spheres, like a dented football. This way, the particles became anisotropic.’
Those building blocks in turn can form larger structures. Meester developed a new method to facilittae this: the colloidal recycling method. ‘We start with loose particles in water. Adding salt makes the particles stick together. However, it is not really possible to control which structures are formed and you can get ugly, unusable structures. We have now devised a technique to convert these aggregates into useful, well-defined forms. We do this by adding a small amount of an oily liquid. It does not mix with water, but it does have affinity for the colloids. These oily droplets act as lubricants between the bulbs, allowing them to glideover each other. When all these small droplets merge into one big drop, you get very compact structures. '
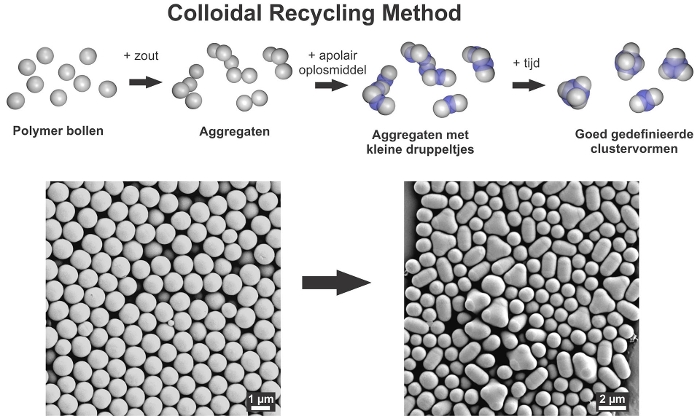
Bulk in a beaker
What is special about this method is the fact that researchers can use it in bulk. Putting the ingredients in a large beaker means that many structures can be made in one go. This is different with traditional methods that work in 2D. The new method can also be applied to many different types of particles.
For Meester, this new method is the most exciting outcomeof her research. ‘It actually surprised us that the method works so easily and that it offers so many possibilities! Sometimes unusable structures also occur automatically, in building blocks that we have created and want to preserve. That is a problem. With this method we can recycle the ugly structures or start with a good base product (stable spheres) and create ourselves the structures we want. That is very cool.'

